Spring 2022

Patient Diversity Campaign
May 2022 campaign
View Content in Digital Newspaper
- Atlanta Voice
- Michigan Chronicle
- La Opinion
- Chicago Citizen
- Philly Tribune
Value Add Digital News Editions
- Chicago Defender
- News Pittsburgh Courier
- Atlanta Daily World
- Atlanta Tribune
Our Patient Diversity Campaign aims to highlight the importance of diversity in clinical studies by building trust and awareness across all populations. We publish a full-page educational advertisement and article in 5 different newspaper publications, print and digital, reaching over 3 million diverse individuals across Black, African American, Hispanic, and Latino communities.
Thank You to the Supporting Organizations
Share Your Voice
To participate in this campaign or an upcoming media campaign, please contact Matt Steele | msteele@ciscrp.org.

Previous Campaigns
Straight Talk on Clinical Trials: Patient Perspectives on Clinical Trial Participation
Mike Wenger of Informa Pharma Intelligence and Wes Michael of Rare Patient Voice and panelists discuss insights from a recent survey of patients on their perspectives of clinical trials, reasons for non-participation in clinical research and ways that sponsors, advocacy organization and recruitment companies can refine the clinical trial enrollment process. Sponsored by Informa Pharma Intelligence and Rare Patient Voice.
View companion material: Q&A | Article Brief | Infographic
About the Panelists
Click on each photo to read biography.
Pediatric Education Series
This 4-part, live-action video series explains clinical research to children and teenagers.




Part 1: Overview of clinical trials
Part 2: Who is involved (research professionals, doctors, nurses, etc.)
Part 3: What it means to consent to be in a trial
Part 4: What treatments are given in clinical trials
This video is also available with Spanish subtitles here and is available in French.
These videos were developed with feedback from patients, parents, and community advocates involved in pediatric research. They all helped to make sure the topics, language, images, and design of these videos are appropriate, educational, and engaging for pediatric audiences.
These videos were also reviewed by an Institutional Review Board (IRB), which is also known as an independent ethics committee. The IRB review ensures the videos follow ethical guidelines for providing information about clinical research to patients and the public.

Silver Awarded in the Category of Digital Health Media / Publications

This video series was done in collaboration with Otsuka
Eye Know—Do You? Podcast with Ken Getz

Bryan and Bradford Manning chat with Ken Getz, a Tufts University School of Medicine professor and founder of CISCRP, a non-profit organization that helps the general public understand the clinical research process, including those living with an IRD. He discusses the benefits and risks of clinical trials, and the value of participation in research.
About Eye Know—Do You? Podcast
Join the Two Blind Brothers, Bradford and Bryan Manning, on a journey to learn more about genetic testing. Diagnosed with Stargardt disease as boys, Bradford and Bryan are known for their advocacy and charitable work in the inherited retinal disease (IRD) community. Eye Know – Do You? is sponsored by Spark® Therapeutics.
To learn more about inherited retinal diseases and the importance of genetic testing visit www.EyeWant2Know.com.
Some speakers have been compensated for their time.

Paying It Forward: Advocating for Others in Clinical Research
CISCRP hosted a 3-part webinar series titled “Navigating Rare Disease and Clinical Research: Every Patient Matters”. This article brief is based on the third episode, “Rare Disease Clinical Trials: After Participation, Paying It Forward”. Moderated by Britta Dornan, Senior Director of Communications and Marketing at EveryLife Foundation for Rare Diseases, panelists included Ryan Colburn, Rare Disease Patient Advocate, Pompe Disease, Dr. Kim Stephens, PhD, President of Project Alive and Marc Yale, Advocacy and Research Coordinator at the International Pemphigus and Pemphigoid Foundation. You can access the webinar recording here.
To “pay it forward” is a philosophy popularized by author Catherine Ryan Hyde’s novel of the same name, where acts of kindness received are not reciprocated to their originator, but forward through the world, creating a ripple effect of good deeds, both big and small. This sentiment is shared by many members of the clinical research community, via the avenues of clinical trial participation, advocacy, and awareness.

“I am a big proponent of paying it forward, sharing your story and experiences for others to benefit. That’s why I created a blog for my rare disease, lymphemadiary.com,” says Britta Dornan, a rare disease advocate, communications professional with EveryLife Foundation for Rare Diseases, as well as a rare disease patient for over three decades. Lymphedema (LE) is a chronic lymphatic disease that can be hereditary, or can occur after a surgical procedure, infection, radiation, or other physical trauma. (1) Despite wearing compression garments 24 hours a day, Britta skis, is a runner and has climbed the Great Wall of China. “I am determined to challenge my limits, rather than let LE limit my challenges,” says Britta.
Marc Yale views becoming an advocate and choosing to participate in a clinical trial as being intertwined concepts. It took six months for Marc to be correctly diagnosed with Cicatricial Pemphigoid, a rare autoimmune skin blistering disease. (2)
“I was kind of angry, I was like ‘How come nobody knows about this disease?’ I understand it’s rare, but shouldn’t doctors know about this? As a patient, you go to the doctor and expect them to be able to answer your questions,” says Marc. In order for more doctors to understand and know about this disease, Marc realized he had to become an advocate for himself and others and create awareness about it.
While seeking a diagnosis, Marc lost the vision in his left eye to the disease. “I was in sort of a desperate situation. I had to take aggressive steps and so I thought, ‘What about a clinical trial?’” recalls Marc. He applied for a clinical trial in France, but he did not meet the inclusion criteria. The experience spurred him on to advocacy efforts.
“What else can I do to advocate for people with my disease so that they don’t have to go through what I was going through? That was the “aha” moment for me, where I decided that I am going to crank disease awareness. I decided that I wanted to become an expert about it, so that when I get into a room with researchers, I know what I am talking about,” says Marc.
He initially volunteered with the International Pemphigus and Pemphigoid Foundation and helped create a peer health coach program, before joining as staff. “I found a community of people that was experiencing what I was experiencing, and it really gave me a lot of hope,” says Marc. He later focused his efforts on the legislative and policy side of advocacy. “Policy and regulation and changing those things is really my calling,” says Marc.
Britta adds, “Working to impact and change policy is a long game. It can sometimes feel like Sisyphus, you’re rolling and rolling and pushing that boulder up the hill, and it keeps rolling back over you down the mountain.” Maintaining focus on the end game of effecting change, is key.
“My son Cole was diagnosed with Hunter Syndrome when he was 2 and a half. Immediately, I was thrown into the world of rare disease, not having any experience in that area at all, but knowing that I needed to find our next steps. We jumped into the clinical trials world,” says Dr. Kim Stephens, PhD. Hunter Syndrome is a rare progressive disease almost always occurring in males, where the body does not break down sugar molecules. Cases range from mild to severe, and life expectancy may be shortened. (3) Cole qualified for the trial, but it was placed on hold for two years. Cole entered the trial at age four and a half and has been participating in the clinical trial for over six years.
Kim’s experience as an executive communications professional strengthened her skills as a patient advocate for her son, as well as others impacted by the disease. Her work includes serving as President of Project Alive, a non-profit organization dedicated to research and advocacy for Hunter syndrome. Her role includes meeting with clinical researchers to provide input about clinical trial design. “I want to make sure that all the clinical trials are successful in our space, because we can’t afford to have any boy in a clinical trial that is going to fail. There’s a real urgency for me to get out there,” says Kim.
“Sometimes, we look around for someone else to step up and then we’re like, ‘But what about me?’” says Kim. “A lot of our motivation is, if we can help someone else not go through what we’ve been going through, then that’s a big plus. As advocates, we’re always there saying ‘Well, what about this, or have you thought about this?’ and thinking more broadly. I’m urging companies now to treat boys that are older, not just doing treatments for newborns through five-year-olds – what about the boys who haven’t been able to be in a clinical trial? Really thinking about how can we include as many patients as possible in a clinical trial.”
“I don’t think I ever thought, even to this day, ‘Oh, I am going to be an advocate’” says Ryan, a patent-holding engineer who was diagnosed with Pompe disease in 2015. Pompe is a rare disease that disables the heart and skeletal muscles. (4) “When I got diagnosed, I tried to learn about the disease, for myself, just so I understood what it was. I continued to figure out that there were a lot more questions that I had that did not have answers, than the ones that did have answers. My mindset about clinical trials is that they’re about participation, and they are also one of the ways we can ask questions and learn something about our disease.” Ryan’s clinical trial participation is now in the double digits.
“Trials can be these long multi-year things, or they can be a day long. Some of them have been as simple as giving a blood sample, so that we can learn new techniques to do newborn screening. Some of them have been traveling regularly to get an experimental treatment injected into your veins. Participation has to make sense for you. It should not be like this sacrificial hero’s aspiration,” says Ryan.
There are tremendous benefits of paying it forward and making contributions that play to one’s individual strengths.
“It was cathartic to be able to do something, and for me, it was part of that healing process, in coming to terms with my condition. That power that I lost when I was diagnosed, I gained back by being able to do something to help others,” says Marc. “You have to ask yourself how can I take this lived experience and make it better for somebody else?”
“I am sort of a political science nerd, so I love going to The Capitol and talking with the Senators and our Representatives. I think of it as I am creating this broad network for myself and for my community, because I don’t know what we’re going to need in the future, and you never know who you are going to influence,” says Kim.
“You don’t have to go all in. It starts by doing something, and you do something a bunch of times, and all of a sudden, you’ve really done something. It doesn’t have to be this big, daunting thing in front of you,” says Ryan.
“Paying it forward is the idea of empowerment and taking back control. We may have these diseases, but they don’t have us,” says Britta.
You can view part-one of the series, “Rare Disease Clinical Trials: Being Informed About Clinical Research” here and part-two, “Rare Disease Clinical Trials: How to Prepare for When the Clinical Trial Ends” here.
Access the companion infographics:
- Clinical Research Participation Roadmap | Part-one here.
- A Garden of Questions to Ask Before the Clinical Trial Ends | Part-two here.
- Paying it Forward in Clinical Research | Part-three here.
To search for medical conditions in a specific location, visit our Search Clinical Trials page.
To stay informed about clinical trials, visit our Resources page.
Sources:
- https://lymphaticnetwork.org/living-with-lymphedema/lymphedema/
- https://pubmed.ncbi.nlm.nih.gov/1869688/
- https://my.clevelandclinic.org/health/diseases/17932-hunter-syndrome
- https://www.ninds.nih.gov/Disorders/All-Disorders/Pompe-Disease-Information-Page
Written by Melissa E. Daley, Communications & Marketing Manager, CISCRP
Working Towards A More Inclusive Environment: Transgender & Non-Binary Participants in Clinical Research
Members of the transgender and non-binary community face myriad challenges during healthcare interactions, including unequal treatment and inappropriate curiosity from healthcare staff. Additionally, there is a widespread lack of understanding of specific transgender health needs and difficulties assessing approaches to healthcare by medical professionals. As a result, 37% of transgender people and 33% of non-binary people responding to a recent survey, reported having avoided medical treatment for fear of discrimination. 62% of transgender people said they have experienced a lack of understanding of specific trans health needs by healthcare staff. (1) A report published by the UCLA School of Law Williams Institute reports that 1.4 million adults in the United States identify as transgender. (2) Colleagues at Parexel International recently conducted a comprehensive review of current medical literature to assess the barriers that transgender and non-binary individuals face to medical care and clinical trial participation, in an effort to enhance the clinical research environment and promote inclusivity.

“We spent quite a lot of time speaking with members of the transgender community and we had a patient advisory council meeting at Parexel where we invited members of the community to speak with us, so that we could listen and learn how we can do better as an industry,” says Rosamund (Roz) Round, Vice President, Patient Innovation Center & Decentralized Trials, Parexel International. Recommendations included strategies for clinical site staff and transgender and non-binary individuals seeking health care in general, as well as through participation in clinical research.
Respecting the Individual
“It’s all about the lived experience of the patient. When we think about health professionals and health professionals who run clinical trials, they have to have an understanding of the transgender community, including things such as pronoun use, life experiences, health practices- all of those things are a foundation for making it a more inclusive environment for all transgender patients,” says Liam Paschall, Global Business Training Partner at Parexel, a transgender man who knows how critical and life-altering inclusive healthcare and clinical trials can be to members of the LGBTQ+ community.
“I think for anyone who is new, (to providing medical care to members of the transgender and non-binary community) the important thing is to know that you might not get it right every time, but it’s important that you learn,” says Roz. “If you use an incorrect pronoun, for example, the important thing is to apologize, move on and do better next time.”
Providing Education & Training for Medical Staff
Liam points to the importance of providing education and training for clinical research and other medical staff. Key topics to cover include the differences between biological sex, sexual orientation, gender identity and gender expression.
“We can do all the work that we can on getting the intake forms right and our systems correct, but if every member of a particular facility, hospital – wherever it is – is not fully educated and trained on how to work with trans patients, it’s not going to change,” says Liam. “It comes down to the individual. Every single person who comes into contact with that patient has to know how to talk to that patient, has to know the right questions to ask, has to understand that they can’t react a certain way, for example, if there is a transgender female, who they feel doesn’t necessarily look the role of being female. It’s all about respect.”
Dr. Sebastian Barr, a licensed psychologist and transgender man who specializes in helping institutions and healthcare providers adopt affirming, inclusive and evidence-based practices in both clinical work and research with members of the transgender community, underlines the importance of cultural humility training and education for staff at every level.
“There is true complexity in the diversity of different biological components that transgender people bring, but what I would remind people of is that there is huge diversity in non-transgender people’s bodies and biologies, as well,” says Sebastian.
Bridging the Trust Gap
There are historical and current reasons and examples as to why there is mistrust of the health care system by transgender and non-binary community members. To bridge this trust gap, Sebastian advises recognizing the diversity within the communities.
“There is a lot more marginalization and oppression of trans women, trans people of color, trans women of color, and non-binary folks,” says Sebastian. “How do we tell the trans community that we have their best interests at heart? We make sure that we are talking to everyone.”
Sebastian also recommends building relationships with community organizations and community members who can facilitate recruitment for clinical research and coordinate advisory boards so that “…you’re not just telling people that you’re trustworthy, but you’re demonstrating that you are and that you have actively considered their participation.”
Modifying Intake Forms
The majority of intake forms in medical settings that Liam has viewed are not representative of the transgender community. “It’s hard to get people to share, in terms of their identity and sexual orientation, when it doesn’t seem like the intake forms and the questions being asked are inclusive of who we are,” says Liam.
“In medical systems, we do a lot of gendering. Everything from records, lab results, the way that we talk about anatomy, in the way that we talk, even casually, about patients. Forms are a really important starting point,” says Sebastian. “Actually integrating what people are telling you about themselves and integrating that with how you and your colleagues are discussing who you are working with is a critical next step.”
Reviewing Protocols
Roz recommends that clinical research professionals conduct protocol review sessions to ensure that there are not items built into the protocol that are inadvertently exclusionary. In a conversation with a member of the scientific community, Roz learned that historically, there had not been transgender and non-binary participation in clinical research because it was not written in the protocol.
“Things evolve and the world changes. The scientific review of protocols to see what is really necessary and the need to enable diversity within protocols, where possible, is really important,” says Roz. “Just because it never has doesn’t mean that it can’t.”
Developing a strategy that includes working with patient advocacy groups, patients and caregivers in the actual design of the clinical trial protocol is key.
Removing Barriers to Participation in Clinical Research
In order to remove barriers to participation in clinical trials for the transgender and non-binary community, Liam advises that clinical research professionals recognize “…things like accessibility, language, confidentiality concerns, make sure we’re not ‘outing’ people, cultural humility training for the staff, sexual orientation and gender identity inclusive intake forms, strategies for public outreach and education, financial reimbursement for expenses, and trans-specific resources in general would be a big help.”
Another step is providing non-gendered spaces in medical facilities. “Creating gender neutral bathrooms is really critical and not challenging, operationally, to do, for example. And it can be a really big difference about how comfortable someone feels or their fear of being revealed,” says Roz.
Involving Community Members as Part of the Health Care Team
“I would recommend that the transgender community not be involved in clinical research where there isn’t an identifiable transgender person involved on the team,” says Sebastian. “I know that sounds severe, but there’s so much risk of harm. Look to see who’s guiding the project and if it will be safe for your involvement.”
Liam adds “There’s a lot of drugs that behave differently from one population to another. So whether there’s a transgender person at the medical facility or not, (medical professionals need to) really understand the differences, at the clinical trial stage, that could leave transgender patients at risk of an avoidable safety issue. Having a diverse and inclusive study population does help to ensure an adequate understanding of how the drugs work and how safe it is across the target population.”
In order to design clinical trials that are truly inclusive, clinical research professionals need to have an understanding of the transgender and non-binary community as a whole. To learn more, access the webinar “Improving Access to and Experiences of Transgender and Non-Binary Patients in Clinical Research” here.
Written by Melissa E. Daley, Communications & Marketing Manager, CISCRP
Access a post webinar Q&A educational resource here.
To search for medical conditions in a specific location, visit our Search Clinical Trials page.
To stay informed about clinical trials, visit our Resources page.
Sources:
(1) LGBT in Britain; Health Report. Stonewell, 2018 and 2015 US Transgender Survey
(2) Report: How Many Adults Identify as Transgender in the United States?, UCLA School of Law, Williams Institute
Clinical Trial Participants’ Selfless Gift to Medicine
 By volunteering to participate in clinical trials of new and experimental treatments, medical heroes give society valuable knowledge about the nature of disease, its progression, and how — and how not — to treat it. Participation always carries some risk, offers some hope for a treatment, and even offers some hope for a cure, but it is likely to bring no direct personal benefit to those who volunteer. As such, medical heroes ultimately give a gift that will benefit future generations of patients.
By volunteering to participate in clinical trials of new and experimental treatments, medical heroes give society valuable knowledge about the nature of disease, its progression, and how — and how not — to treat it. Participation always carries some risk, offers some hope for a treatment, and even offers some hope for a cure, but it is likely to bring no direct personal benefit to those who volunteer. As such, medical heroes ultimately give a gift that will benefit future generations of patients.
Facing the challenge
For the vast majority of people, clinical trials are an abstract concept that have little to no personal relevance. Most people take a hard look at clinical trials for the first time when facing the sudden and often unexpected prospect of a serious and debilitating illness for which no medication is available or adequate. Typically, patients, their families, their friends, and their healthcare providers must gather information quickly to understand how the clinical trial process works, the requirements of participation as defined by the study protocol, and whether participation is appropriate. This rush to navigate and master the unfamiliar terrain of clinical trials invariably feels overwhelming and confusing, and once in a clinical trial, many volunteers find the requirements and demands of participation difficult to manage.
Raising awareness
In 2003, the Center for Information and Study on Clinical Research Participation (CISCRP) was founded to provide outreach and education to those individuals considering participation in clinical trials and their support network. Based in the Boston area, this nonprofit organization serves the global community of patients and the public by raising awareness and providing education about clinical trials and by enhancing study volunteer experiences during and after clinical trial participation. Our many events and services are designed to improve public and patient literacy; to engender feelings of empowerment and control; to ensure more informed decision-making, improved convenience, and accessibility; and to recognize and appreciate the medical heroes that inspire us. This special USA Today supplement also plays an important part in raising awareness and literacy. It is a reference resource offering an introduction to clinical trials and providing valuable information to help you and your support network navigate the participation process.
At the present time, nearly 6,000 experimental drugs and therapies are in active clinical trials, and that number continues to grow as improvements are made in detecting disease, discovering new medical innovations, and understanding and addressing the root cause of acute and chronic illnesses. At the very heart of all of this promising, lifesaving, and life-altering activity are medical heroes to whom we owe our deepest appreciation for their profound gift of participation.
Written by: Ken Getz | Founder and Chairman, CISCRP
Article from 2021 Clinical Trials Supplement, USA Today. View Supplement Here >
An Inside Look at Advocating for Yourself or Your Child in a Clinical Trial
 Dr. Mark Sorrentino, MD, MS, Vice President for Pediatric Development, ICON and Dr. Joanna L. Perkins, MD, MS, Director of Medical Affairs, Hematology-Oncology, Americas, ICON, discussed how to advocate for yourself or your child in a clinical trial in a CISCRP 15-minute Flash webinar. Dr. Sorrentino shared his perspective as a clinical trial participant and Dr. Perkins expanded on her experience from a principal investigator’s lens. You can view the webinar here.
Dr. Mark Sorrentino, MD, MS, Vice President for Pediatric Development, ICON and Dr. Joanna L. Perkins, MD, MS, Director of Medical Affairs, Hematology-Oncology, Americas, ICON, discussed how to advocate for yourself or your child in a clinical trial in a CISCRP 15-minute Flash webinar. Dr. Sorrentino shared his perspective as a clinical trial participant and Dr. Perkins expanded on her experience from a principal investigator’s lens. You can view the webinar here.
It was near Thanksgiving in 2020, and Dr. Mark Sorrentino noticed a small lump in his neck which was diagnosed as head and neck cancer. “I found out I had cancer that required surgery, radiation and chemotherapy,” Dr. Sorrentino recalls. “At the time of my diagnosis, my care team told me there was a clinical trial that I could participate in. Given that I had been in clinical research for over 20 years, it was the first time I had the opportunity to participate in a clinical trial – I was both nervous and excited about seeing what it would be like from the other side.” The experience reinforced the importance of being an advocate for oneself or one’s child in Dr. Sorrentino’s mind.
Reviewing Informed Consent & Schedule of Assessments
Starting from the beginning of Dr. Sorrentino’s journey, he received an informed consent document that was 15 pages long. While it was not the longest document of this type that he had seen during his career in clinical trials, it was still somewhat daunting. Dr. Sorrentino thought about how this same document would need to be translated into an assent document for a child. (An assent form differs from an informed consent form and is designed to indicate that a child is willing to participate in a clinical study and understands what he or she will need to do. You can learn more here).
“It was filled with all sorts of legalese, and one thing that I picked out that I thought was very challenging, was the schedule of assessments,” says Dr. Sorrentino. The schedule outlines the treatment, tests and other procedures that may occur during the course of the clinical trial. The documents are not always presented in an easy-to-follow format. Because of his experience in clinical research, Dr. Sorrentino knew to ask for a version of the document that outlined the details in a table.
“Informed consent documents can be completely overwhelming,” says Dr. Joanna L. Perkins. “From a regulatory standpoint, there is a lot of information that has to be included.” And, at the beginning of the study, it’s important to find out how the results of the clinical study will be shared with participants.
It is also important to understand before deciding to participate, what is particularly being done for the trial and what is above and beyond that is specific to that clinical trial and not part of standard of care. This is not always clear for the participant and important to feel confident to ask the study team questions early on. For example, a weekly blood draw that is outlined in the schedule of assessments may feel burdensome, but it may also be part of the regular standard of care for the disease or indication.
Coordinating Care & Providing Support
“One time I went in for my standard of care blood draw, which was in one laboratory area in the hospital. Later that day, when I went to the clinic to do the schedule of assessments for the clinical trial, and I had to have another blood draw,” said Dr. Sorrentino. He asked the clinical study coordinator why a single blood draw had not been scheduled and coordinated, to save time and physical discomfort. “This was an example of the health care teams not communicating with each other,” says Dr. Sorrentino.
“Depending on the clinical trial that you are in, and the center you are at, there may be two totally different teams of medical professionals who are overseeing your clinical care and your study assessments,” says Dr. Perkins. “It’s really important to advocate for yourself or your child to make a point of understanding what is being done on each of the days you go for your visit. Painful procedures are often a part of treatment. Make a point to ask what will be done to support your child through those painful procedures – and adults, as well.” Many clinical research centers have dedicated child-life staff to ensure that pediatric participants are cared for in the most sensitive way to minimize pain and fear. This can include play-based distractions, medical procedures such as sedation, or meetings with pediatric mental health professionals.
Parents may have to coordinate childcare options for siblings, in tandem with scheduling medical treatment for the child participating in clinical research. “Most large pediatric clinical centers have sibling play areas,” adds Dr. Perkins. Other services may include programs that address how to cope when one’s brother or sister is going through treatment for a chronic illness, such as cancer. “Parents should ask about programs to support their other children,” advises Dr. Perkins. Sometimes, there is financial support available for childcare and transportation.
Coordinating visits to the clinical research center with standard of care visits, when possible, can greatly reduce the burden of clinical research participation for both adults and children. It is important to advocate for yourself and/or child when scheduling clinical trial visits with the study team. “Take a really good look at when you have to be at the clinic,” says Dr. Sorrentino.
Bringing the Clinical Trial to You
Remote capabilities are being scaled in clinical research, due, in part, to the advent of the COVID-19 pandemic. Some tests may be able to be performed at home with the use of portable medical devices and apps, coordinated through a laptop, tablet or mobile phone. Clinical research staff, including nurses, can often make home visits. Dr. Sorrentino was able to be evaluated at home, outside in his yard, during the COVID-19 restrictions in his hometown of Los Angeles. “It was fantastic, it kept me from having to go down to the hospital,” says Dr. Sorrentino. “The nurse took my vitals and did my lab work.”
Asking Questions About the Placebo
Another point of advocacy is around the use of placebos. Inquire what happens at the end of the study. If your child was on the placebo, ask if they will be able to receive the therapeutic drug or agent that was being studied at that point of time. “Many times, that is the case, but you have to remember to ask about it,” says Dr. Sorrentino.
Developing New Treatments & Optimizing Care
Overall, Dr. Sorrentino views his experience as a clinical trial participant in a positive light. “Clinical trials need you and they need your children, if we’re going to get new and innovative therapies, particularly for children, since we have lagged behind on that in the past,” says Dr. Sorrentino. It’s important to remember there is a whole team of medical people watching your disease and diagnosis.
“The overall cure rate for pediatric cancers in the 1960s was about 20%,” says Dr. Perkins. “And now, today, it’s higher than 80%. The biggest, most important reason for that is the sharing of data and participation in clinical trials. With standardized clinical trials, we can learn much more quickly how to optimize care and increase survival rates for diseases like cancer,” says Dr. Perkins.
Authored by: Melissa E. Daley, Communications & Marketing Manager, CISCRP
View CISCRP’s library of webinars and podcasts here.
To search for medical conditions in a specific location, visit our Search Clinical Trials page.
To stay informed about clinical trials, visit our Resources page.
Supporting Caregivers in Clinical Research During the Pandemic
The COVID-19 pandemic has escalated the social isolation, medical uncertainty and disruptions in home/work life that rare disease patients and caregivers already experience on a daily basis. “We’re 18 months into a chronic grind of events that might not be impacting us dramatically, day to day, but there’s this cumulative, chronic, wearing-downness,” observes Scott Schliebner, former Senior Vice President, Scientific Affairs and Therapeutic Expertise Head, Center for Rare Diseases, ICON. Participants in clinical studies are surrounded by a support system of caregivers that help facilitate their participation. In the rare disease space, this often means parents. Great strides have been made in the clinical research enterprise to scale patient-centric approaches. This spectrum of support is also being extended to caregivers of participants in clinical research.
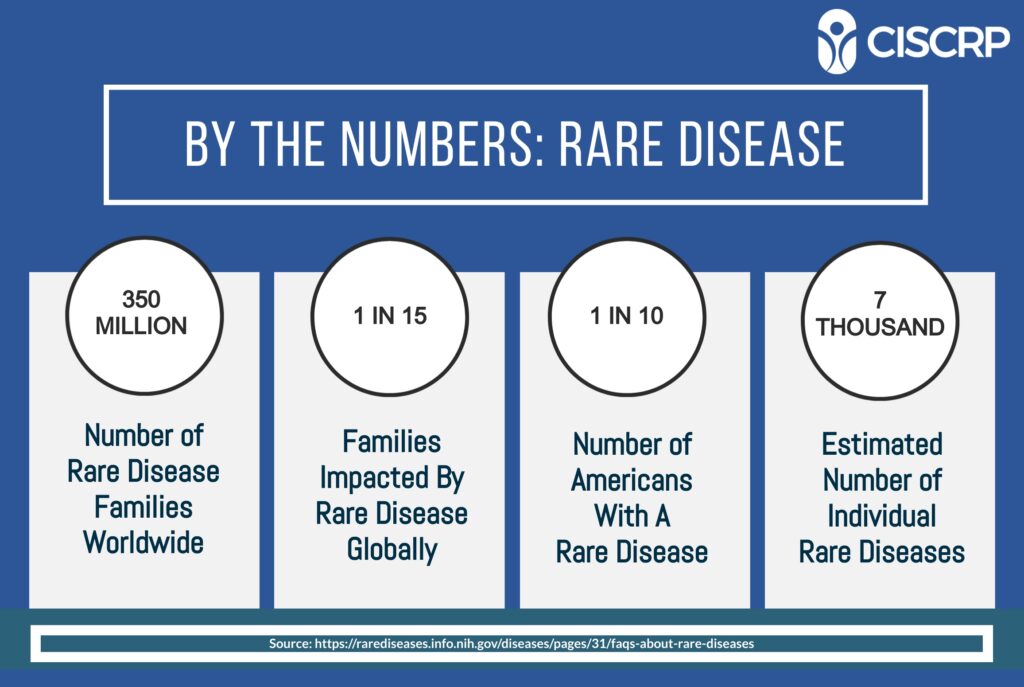
“Half the patient population in rare disease is children. The caregiver support is incredibly important. These are the individuals making decisions on behalf of their child, or their patient-adult child, it’s not all under 18 minors. Many of these patients have grown into adulthood and need ongoing support. Parents are involved in clinical trial decisions, advocacy decisions, and treatment decisions,” says Cristol Barrett O’Loughlin, Founder and CEO of Angel Aid Cares, a non-profit that provides mental health and wellness services to rare families,
particularly caregivers, with a focus on rare disease mothers. Offering assistance at different levels to caregivers is essential, particularly when it comes to emotional support.
In a study published by Health Psychology, rare disease patients were recently surveyed and interviewed about the types of support they receive, stress levels and their overall satisfaction with life. Four types of support were evaluated by the respondents:
- Emotional support where the individual feels understood and validated by others
- Informational support where individuals receive advice from others
- Tangible support which provides practical assistance to address day to day tasks
- Companionship support where individuals develop a sense of belonging by participating in leisure activities with others
Emotional and companionship support were the strongest predictors for life satisfaction, regardless of stress levels, with companionship being rated as the most significant factor. (1)
Cristol explains that these types of support can also be extended to the caregiver experience.
“Informational and tangible support are the triage list of what has to get done. What is this disease? What are the symptoms? What doctor do I go see to treat those symptoms?” says Cristol. “Are there any clinical trials? How do I get enrolled? How do I get insurance coverage for my claims? What medical devices do I need to have at home to manage this experience for my patient-child or patient-adult? Emotional support and companionship is where that caregiver or patient feels that someone understands their feelings about what’s happening to them. Someone is really, truly listening and engaged at a heart level, with what they’re experiencing.” This emotional and companionship level is enhanced further if the other person is currently undergoing or has undergone the same experience, providing a sense of relief for the patient or caregiver.
Cristol knows from personal experience the toll that stress takes on caregivers. She assisted with caring for her three older brothers who were born with a rare genetic disease and passed away at ages 12, 18 and 19 years old. “For decades I witnessed this chronic stress within the family, with caring for healthy individuals in the family and medically fragile individuals within the family,” says Cristol. “This isn’t a stressful event, as in PTSD, and now we’re recovering from it. This is ongoing, chronic stress.”
Cristol shares her insights into what she terms as CTSD, or chronic traumatic stress disorder.
“In a stressful event, your cortisol spikes and you go into a heightened awareness state and then you go into recovery (PTSD). (2) But when you are a caregiver, you elevate into this stressful state and now you’re buzzing at that higher level. You are in this for the long haul. The relief that you bring to CTSD or to a caregiver is different than trying to recover from a situational traumatic experience. That relief comes in the form of daily micro-moments of self-care,” says Cristol. This moment can be as simple as implementing slow breathing techniques to lower the heart rate. “Breathwork is the number one thing we can do,” says Cristol. Other wellness methodologies to implement include healthy eating, sleeping and exercise habits, all of which can be incredibly challenging for caregivers to navigate.
“Mothers are the primary caregiver in 82% of families in the rare disease space,” says Cristol. “We know (from support provided by Angel Aid Cares) that focusing on a mother, a grandmother, or an aunt, within a family, we can uplift a whole family. It’s never an exclusion of any other member of the family, rare fathers included.” Cristol also makes note that there is much to be learned from the mature caregiving models of the veterans, military, senior and dementia care spaces.
“If you want patients to enroll in a clinical trial, we need to make things easier for them, and part of this, is, of course, just extending this concept of patients to caregivers. What else can we do with that care circle to help that whole family unit or care circle group participate in a clinical trial? There are a lot of practical things,” says Scott.
To learn more, watch the 15-minute Flash webinar “Supporting Caregivers in Clinical Research: Navigating the Pandemic” here.
Authored by: Melissa E. Daley, Communications & Marketing Manager, CISCRP
To search for medical conditions in a specific location, visit our Search Clinical Trials page.
To stay informed about clinical trials, visit our Resources page.
Sources:
(2) https://www.psychiatry.org/patients-families/ptsd/what-is-ptsd














































