From "The Gift of Participation" by Ken Getz, Founder & Board Chair, CISCRP
Payment for participation is considered compensation for your time and commitment to comply with the protocol. Healthy volunteers in phase I studies of a new treatment or device are usually paid for their participation. Study volunteers in phase IV studies of drugs already on the market often receive compensation. And payments in clinical trials are often given to study volunteers when the health benefits of an experimental treatment or new device are not known or are remote.
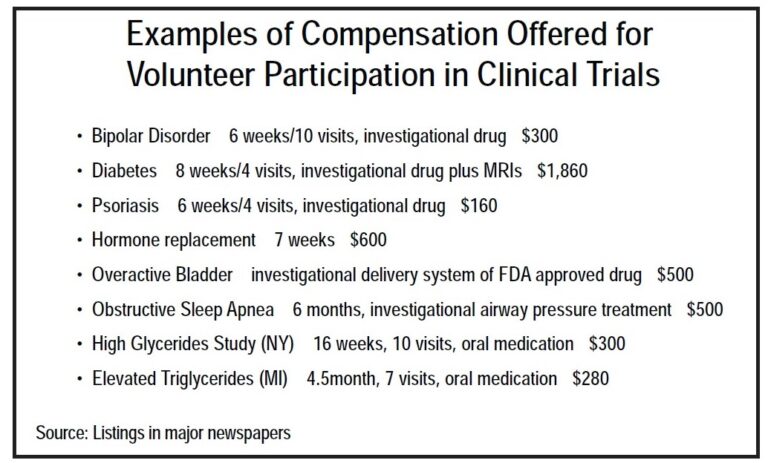
The compensation amount varies widely and depends on many factors including the length of the clinical trial, the number of visits that you’ll need to make during the study and the number and types of procedures that will be performed. Compensation can range from a hundred dollars to several thousand dollars. Partial payments are made over the course of participation. The amount and schedule of payments is proposed by the sponsor of the clinical trial to the IRB overseeing the study. The IRB will review, and in some cases revise, the proposal to ensure that it does not coerce or influence someone to agree to participate. Patient recruitment advertisements usually list compensation amounts. The informed consent document lists compensation amounts and payment schedules. You can also ask whether a trial is offering compensation and the amount when calling a research center to inquire about participation.
Once participation begins, the research center typically writes each volunteer a check as the study progresses—usually on a per-visit basis. A small bonus may also be paid to volunteers who complete the entire study. In some cases, study doctors may dispense approved medication samples and provide additional care free of charge after the trial to show their appreciation to study volunteers.
Any financial compensation paid to study participants is considered taxable income by the Internal Revenue Service (IRS). However, research centers are obliged to report to the IRS only payments to individuals that total $600 or more for the year. These Form 1099s for “Miscellaneous Income” are generally sent out to participants in January, the same month employers are shipping out W-2 forms to their employees.
If a volunteer decides to withdraw from the study before completing the trial, the research center will usually provide partial compensation.
For more information on clinical trials and making informed decisions about volunteering for clinical research, read “The Gift of Participation” by Ken Getz, Founder and Board Chair, CISCRP.
You can find the book here.
To search for medical conditions in a specific location, visit our Search Clinical Trials page.
To stay informed about clinical trials, visit our Resources page.





 “Race, literacy, financial barriers, access, social, systemic and hereditary backgrounds should not be determinants of life and health equity,” said Maimah Karmo, President, Tigerlily Foundation.
“Race, literacy, financial barriers, access, social, systemic and hereditary backgrounds should not be determinants of life and health equity,” said Maimah Karmo, President, Tigerlily Foundation.

 Fifteen years ago, at the age of 42, Israel Robledo was diagnosed with Parkinson’s disease. Shocked, he spent the next nine months coming to terms with his diagnosis. “I was married. I had three girls at home. I was working as a schoolteacher. And I was thinking, ‘Oh my gosh, my life is over,’” he says. “I only knew about older people having Parkinson’s.”
Fifteen years ago, at the age of 42, Israel Robledo was diagnosed with Parkinson’s disease. Shocked, he spent the next nine months coming to terms with his diagnosis. “I was married. I had three girls at home. I was working as a schoolteacher. And I was thinking, ‘Oh my gosh, my life is over,’” he says. “I only knew about older people having Parkinson’s.” Clear Messaging
Clear Messaging