The Michael J. Fox Foundation for Parkinson’s Research (MJFF) aims to speed clinical research by removing obstacles that stand in the way of drug development. In pursuit of this mission, the Foundation gathers insights from a wide range of stakeholders and uses these perspectives to enhance clinical trial processes from start to finish. In Applied Clinical Trials’ Eye on Patient Advocacy series, we will share best practices and lessons learned from the field of Parkinson’s research that can be applied to clinical trials across disease states. In our fourth column in this series, we explore retention strategies used in a landmark longitudinal Parkinson’s disease (PD) study.
To complete a study, it is critical to retain study participants. Participant attrition has the potential to interfere with the scientific validity of a study and distort data designed to measure drug efficacy and safety. According to Forte Research:1
- Eighty-five percent of clinical trials fail to retain enough participants.
- The average dropout rate across all clinical trials is 30%.
Patient retention is an important element of the Parkinson’s Progression Markers Initiative (PPMI), a landmark, longitudinal, observational study sponsored by The Michael J. Fox Foundation. PPMI (ppmi-info.org) aims to find reliable and consistent biomarkers for PD progression by studying cohorts of Parkinson’s patients (de novo idiopathic PD and PD-manifesting genetic mutation carriers), populations at risk for PD (non-manifesting genetic mutation carriers and subjects at risk due to REM sleep behavior disorder or hyposmia), and controls without PD.
Participants in PPMI commit to long-term participation, providing biospecimens (e.g., blood, urine, spinal fluid), and undergoing multiple neuroimaging, clinical and behavioral procedures, and assessments over a period of at least five years.
The study launched in 2010, and since then, approximately 1,500 individuals have enrolled. PPMI’s retention rate has consistently held strong, year after year, at about 86%.
To ensure steady participation and to prevent attrition, PPMI weaves together four key tenets of retention, cultivated and refined since study launch: 1) facilitate participation; 2) communicate study progress; 3) express appreciation; and 4) inform participants of study results.
Facilitate participation through travel concierge services, reimbursement, and remote visits. PPMI study leadership prioritized and simplified long-term participation in large part because individuals carrying specific PD-linked genetic mutations live across a wide geographical area. To facilitate volunteers’ continued participation, PPMI cultivated a boutique experience for them and their care partners. Prospective and enrolled PPMI participants are given the option for complimentary roundtrip transportation between their home and appointments at two “super sites” that have the capacity to handle a high volume of study volunteers.
A third-party vendor manages all logistical planning, including participants’ accommodations, meals, and travel to and from study visits. This door-to-door service reflects the value PPMI study leadership puts on participation and participants. For participants who choose not to travel to a super site, travel and hotel expenses are reimbursed through Greenphire/ClinCard. PPMI leadership is always looking for ways to reduce participant burden and is currently exploring the possibility of remote (video and enhanced phone) and home visits.
Communicate study progress through newsletters, update calls, and a centralized webpage. Reminding participants of the bigger picture is a meaningful way to engage them in the collective success of a study. According to a 2017 report by The Center for Information and Study on Clinical Research Participation,2 the number one reason individuals choose to participate in clinical research is to help advance science or the treatment of a disease or condition. Given this initial motivation, updates on study progress and contributions to the field will facilitate continued engagement. In PPMI, study progress is communicated in several ways:
- PPMI newsletters provide high-level updates on the study (e.g., study enrollment progress, how the data and samples collected are being used for research) as well as interviews or profiles of study participants and/or study staff.
- PPMI update calls, which are scheduled throughout the year, feature presentations and Q&A sessions with study researchers and study team members.
- A PPMI participant webpage allows centralized access to digital versions of the participant newsletters and recordings of study update calls.
- PPMI blog provides regular news about the study, including recent findings that have emerged from the data.
Express appreciation through a thank-you booklet. Letters from members of the Parkinson’s community, researchers, MJFF staff, statisticians, and study coordinators were published in a print and digital booklet to thank and honor volunteers for their participation. Collecting the personal reflections of the many individuals involved in or impacted by PPMI is a meaningful way to empower participants and remind them of the larger cause they are tied to.
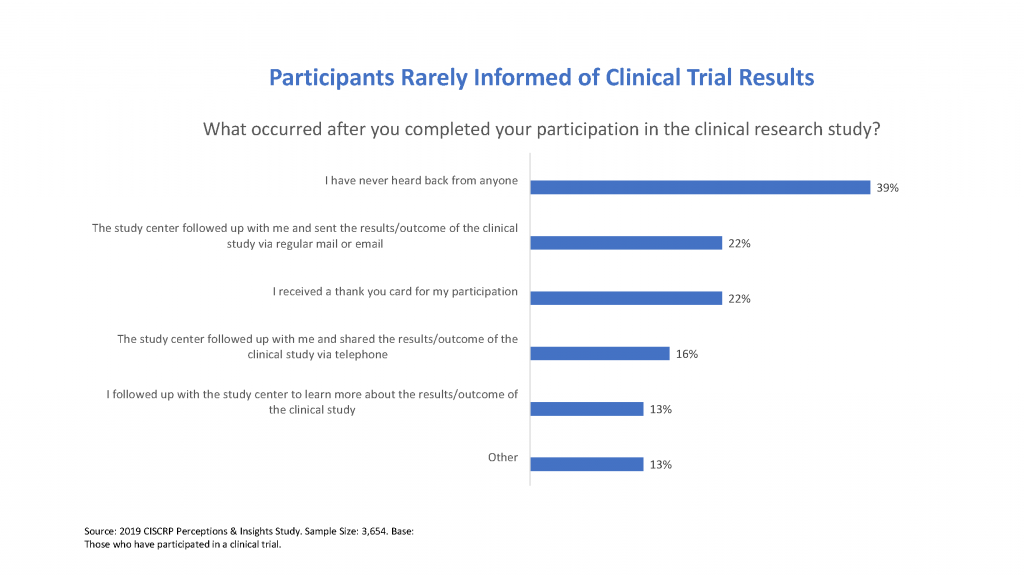
Inform participants of study results through newsletters, update calls, and a webpage. The majority of study volunteers (90%) want to receive results from the clinical trial in which they participated.3 Because there is ongoing analysis of PPMI data and continued follow up of participants, study results are shared on a rolling basis. Using familiar channels to communicate study progress is a great way to close the loop with study participants.
Putting it all together: Host an event
PPMI staff and study leadership also show their commitment to the study’s success, and their appreciation for participants, by hosting annual study update luncheons and dinners that incorporate all the tenets of retention. Having an in-person get together gives participants the chance to meet other volunteers and share experiences of living with PD and taking part in PPMI. During these events, local site staff present study progress and provide relevant results from ongoing data analysis. Michael J. Fox Foundation staff also attend and, together, all study stakeholders thank participants for their time and commitment.
Data used in the preparation of this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (www.ppmi-info.org/data). For up-to-date information on the study, visit www.ppmi-info.org.
PPMI—a public-private partnership—is funded by The Michael J. Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Allergan, Avid Radiopharmaceuticals, Biogen, BioLegend, Bristol-Myers Squibb, Celgene, Denali, GE Healthcare, Genentech, GlaxoSmithKline, Lilly, Lundbeck, Merck & Co., Meso Scale Discovery, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi Genzyme, Servier, Takeda, Teva, UCB, Verily, and Voyager Therapeutics.
References
- Lopienski, Kristina. “Retention in Clinical Trials – Keeping Patients on Protocols.” Forte Research. June 1, 2015. https://forteresearch.com/news/infographic/infographic-retention-in-clin…
- The Center for Information and Study on Clinical Research Participation (CISCRP). “2017 Perceptions & Insights Study: Public and Patient Perceptions of Clinical Research.” https://www.ciscrp.org/wp-content/uploads/2019/06/2017-CISCRP-Perception…
- Getz, Kenneth. “Events That Have Shaped Study Participant Protection.” The Gift of Participation: a Guide to Making Informed Decisions about Volunteering for a Clinical Trial, 2nd ed., CISCRP, Center for Information & Study On Clinical Research Participation, 2014, pp. 158–158.
James Gibaldi, MS, Associate Director; and Bernadette Siddiqi, MA, Associate Director; both with The Michael J. Fox Foundation in New York, NY. To contact the MJFF Recruitment and Retention Team, email: trialsupport@michaeljfox.org
MJFF would like to acknowledge the following individuals for their contribution to the research presented in this case study: Vanessa Arnedo, MPH, Sarah Berk, MPH, Sohini Chowdhury, MA, Tara Hastings, MA, Catherine M. Kopil, PhD, and Alyssa Reimer.